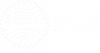Olopatadine
The recurrent attacks of allergic diseases such as allergic conjunctivitis and urticaria are common medical needs that affect the quality of life of more than one billion people worldwide. Antihistamines are the first-line treatment for such diseases, and olopatadine is a representative variety among them with both high potency and high safety. As a second-generation H1 receptor antagonist, it can simultaneously inhibit the release of inflammatory mediators from mast cells and antagonize histamine receptors. Its dual mechanism of action greatly reduces central adverse reactions such as drowsiness. Clinically, it can be formulated into eye drops to relieve symptoms such as eye itching and conjunctival congestion caused by allergic conjunctivitis, and its oral dosage form can also be used to treat urticaria and allergic rhinitis, with the applicable population covering children over 3 years old and adults.
The global market size of olopatadine API was approximately USD 120 million in 2023, and it is expected to expand steadily at a compound annual growth rate of 5.8% by 2028. In terms of the domestic market, after the patent of the original research enterprise expired, domestic generic drugs have rapidly increased their volume, and the current localization rate has exceeded 75%. East China and South China are the major domestic production clusters of APIs. In the local volume-based procurement since 2021, the average bid price of oral olopatadine preparations has dropped by 62%, which further drives the continuous release of demand for upstream APIs.
CATO can provide a full set of impurity reference standards for olopatadine API. All products are in stock and meet the quality requirements of multiple regulatory systems such as Chinese Pharmacopoeia and FDA, and can provide stable and reliable reference standard support for pharmaceutical enterprises in such work as raw material quality research and generic drug consistency evaluation.